Department Environmental Chemistry
Environmental Chemistry Stable Isotope Lab
The research group of the Environmental Chemistry Stable Isotope Lab studies how organic chemicals react in natural and engineered environments.
Our goal is to provide the fundamental knowledge on chemical mechanisms of transformation that enables one to rationalise observations of pollutant dynamics and describe interactions of organic compounds in biological and abiotic systems quantitatively. We specifically aim at bridging the interface between basic and user-inspired research questions by applying process-based information to assess the fate of chemicals in diverse environmental settings ranging from contaminated soils and sediments to water treatment facilities.
Our scientific approach combines the experimental study of the rates and pathways of
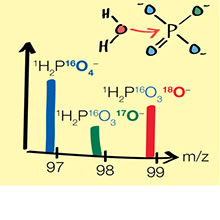
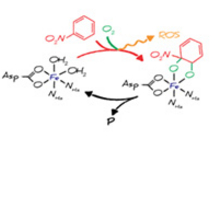
(bio)chemical reactions of organic pollutants from the molecular to the field scale with specific characterization of properties and reactivities of the environmental phases such as enzymes and minerals which catalyze these processes. The tools applied in our studies cover different types of mass spectrometry to quantify the kinetics, isotope effects, and product of (bio)chemical reactions, electrochemical and spectroscopic techniques for the description of apparent thermodynamic properties of minerals, as well as quantum-chemistry based computations to reconcile theory with experiment.