Department Environmental Chemistry
CyanO3: cyano-metabolites from source to tap
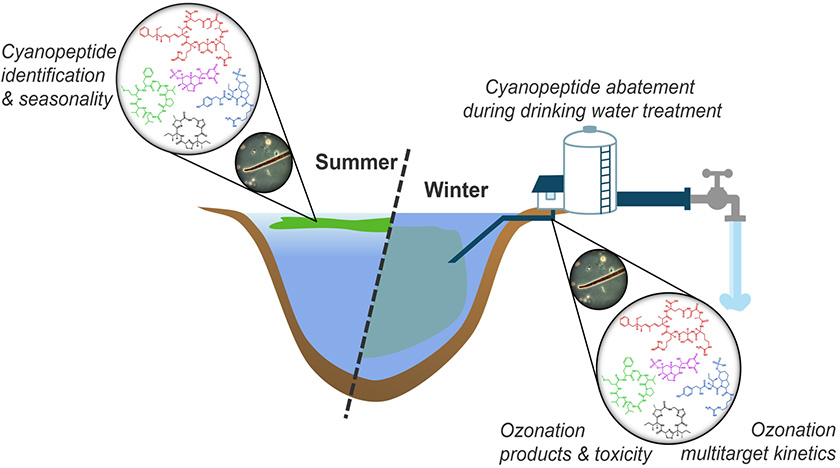
Cyanobacterial bloom events are frequently observed in freshwater resources across the globe and produce known toxins. Microcystins, one class of cyanopeptides, have been studied intensively and the wealth of evidence regarding exposure concentrations and toxicity led to their inclusion in the World Health Organisation water quality guidelines. However, cyanobacteria produce a large diversity of cyanopeptides beyond this class of microcystins. The question arises, whether the other cyanopeptides are of human and ecological concern.
We are currently working on cyanopeptides “from source to tap” in collaboration with the water works Zürich (Wasserversorgung Zürich, WVZ). Here we ask the questions, whether these compounds reach the drinking water treatment plants and whether the latter are abated to prevent cyanopeptides form reaching the consumer's taps.
Focus of this project:
- Investigating seasonal cyanopeptide profiles in Lake Zurich (the source water) and at the intake to the drinking water treatment plant
- Assessing abatement of cyanopeptides during water treatment
- Determining second-order rate constants for the reaction of cyanopeptides with ozone in novel multitarget method
- Evaluating potential toxicity of transformation products
Ozonation is widely used in Switzerland as an efficient barrier against microorganisms, taste and odour compounds and micropollutants. Ozone can also degrade known cyanotoxins such as microcystins, and thus is a desired process in treatment plants affected by cyanobacterial blooms. However, little is known about the efficiency of ozone for other cyanopeptide classes. This project aims to fill the knowledge gap on the reactivity of a large variety of cyanopeptides, transformation product formation, and evolution of cyanopeptide-induced toxicity during ozonation. A better understanding of these three aspects will advance our ability to predict their fate during water treatment and to assess potential risks.
