Department Environmental Chemistry
Enzyme Mechanisms and Kinetics of Organic Contaminant Oxygenation
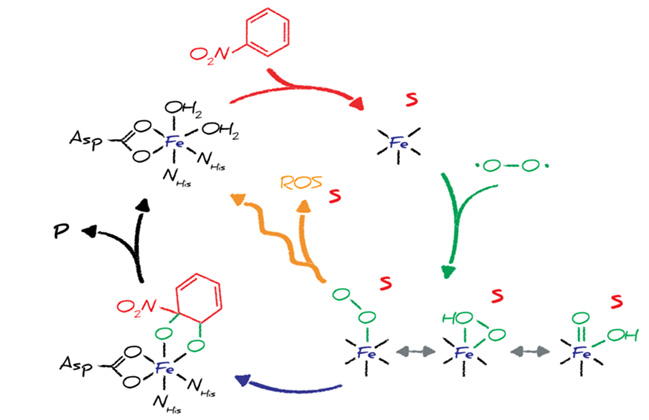
Rieske non-heme ferrous iron oxygenases are the main enzymes capable of hydroxylating persistent (poly)cyclic chlorinated and nitrated hydrocarbons. This class of enzymes therefore plays a critical role in the biodegradation of many recalcitrant organic contaminants in natural environments including soils, groundwaters, and surface waters or in engineered systems such as sewage treatment plants. While the general mechanisms of molecular O2 activation (and formation of reactive electrophilic oxygen species) are well known, essential factors of the catalytic activity of Rieske oxygenases with relevance for biotransformation of complex mixtures of contaminant remain elusive.
In this research, we explore the substrate- and enzyme-specificity of selected Rieske oxygenases with regard to path of molecular O2 activation as well as the efficiency of substrate hydroxylation vs. formation of problematic reactive oxygen species from the unproductive O2 uncoupling. Through the combination of experimental, analytical, and computational approaches, we aim at elucidating the reactivity of this class of enzymes towards the large number possible substrates and provide a mechanistic basis for hypothesis that link O2 uncoupling to the evolution of catalytically more proficient Rieske oxygenases.
