Department Environmental Chemistry
Natural Toxins
Our ecosystems and drinking water resources are not only vulnerable towards anthropogenic pollutants. Natural toxins present an additional threat for which we still lack comprehensive risk assessment and management plans. Among the natural toxins from various kingdoms, those produced by aquatic organisms, such as cyanobacteria have a direct entry into our water resources.
Cyanobacterial bloom events conquered freshwater resources across the globe, yet the potential risk of many cyanobacterial metabolites remains mostly unknown. Only microcystins, one class of cyanopeptides, have been studied intensively and the wealth of evidence regarding exposure concentrations and toxicity led to their inclusion in water quality guidelines. However, cyanobacteria produce an large diversity of cyanopeptides beyond the class of microcystins. The question arises, whether the other cyanopeptides are in fact of no human and ecological concern or whether these compounds merely received (too) little attention thus far.
We focus specifically on:
- advancement of analytical workflows to identify and quantify emerging toxins and bioactive metabolites
- co-production dynamics of toxins and metabolites by bloom-forming cyanobacteria
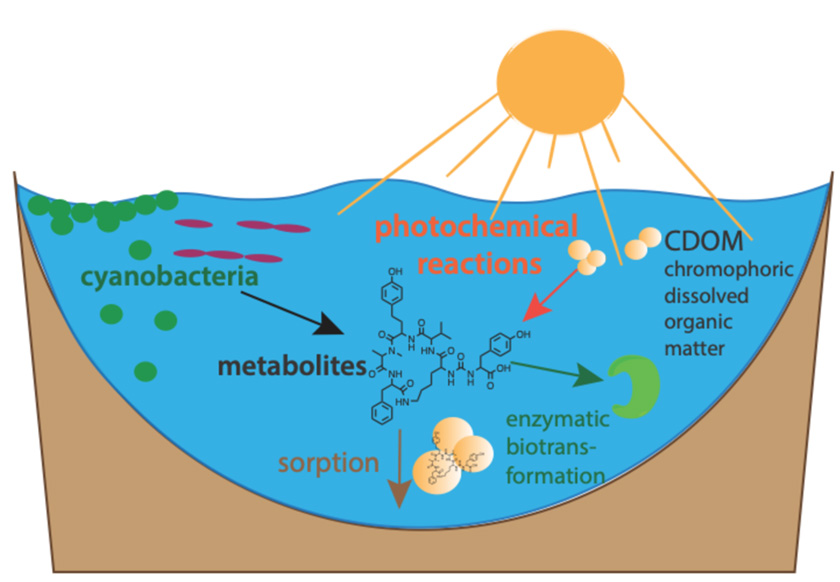
- environmental fate processes of toxins in surface waters and engineered systems
- adverse lethal and sublethal effects towards aquatic organisms
More information on current projects:
Publications
array(2 items) publications => '35348,35323,33271,33380,33003,33024,31895,24660,26097,23735,23739,22156,2231
0,22112,20660,20604,18207' (101 chars) libraryUrl => '' (0 chars)
array(17 items) 0 => Snowflake\Publications\Domain\Model\Publicationprototypepersistent entity (uid=35348, pid=124) originalId => protected35348 (integer) authors => protected'Wang, X.; Ingold, A.; Janssen, E. M. -L.' (65 chars) title => protected'Biotransformation dynamics and products of cyanobacterial secondary metaboli
tes in surface waters' (97 chars) journal => protected'Environmental Science and Technology' (36 chars) year => protected2025 (integer) volume => protected59 (integer) issue => protected'38' (2 chars) startpage => protected'20726' (5 chars) otherpage => protected'20737' (5 chars) categories => protected'microcystins; anabaenopeptins; transformation products; reaction pathways; h
armful bloom' (88 chars) description => protected'Cyanobacteria produce toxic and bioactive secondary metabolites, posing risk
s to ecosystems and human health, yet their transformation pathways in surfa
ce waters remain unclear. We assessed biotransformation for 40 cyanopeptides
including microcystins, anabaenopeptins and cyanopeptolins in surface water
s and <em>in situ</em> enriched biofilm suspensions. In surface waters, most
cyanopeptides did not degrade significantly over the course of 7 days. A wi
de range of biodegradability across cyanopeptides was apparent in biofilm su
spensions from three rivers. Increasing the biofilm density shortened the la
g time and increased initial removal of cyanopeptides. Increasing the initia
l cyanopeptide concentration lengthened the lag time and decreased their ini
tial removal, supporting inhibitory effects of cyanopeptides toward enzymes
involved in their own transformation. Transformation kinetics and product an
alysis demonstrated a structure–reactivity relationship across and within
cyanopeptide classes. Anabaenopeptins were hydrolyzed at the C-terminus when
arginine, tyrosine and (iso)leucine were present, but not when phenylalanin
e or tryptophan was present. Microcystins showed tetrapeptide formation when
adda linked to arginine but not when it linked to alanine, leucine, or tyro
sine. Oxidation of tyrosine and deamination of arginine residues showed an i
nterdependence across cyanopeptide classes. These novel insights into biotra
nsformation products and pathways of a wide range of cyanopeptides facilitat
e assessment of exposure scenarios in surface waters and inform about kineti
cs and product formation in biological water treatment.' (1651 chars) serialnumber => protected'0013-936X' (9 chars) doi => protected'10.1021/acs.est.5c09247' (23 chars) uid => protected35348 (integer) _localizedUid => protected35348 (integer)modified _languageUid => protectedNULL _versionedUid => protected35348 (integer)modified pid => protected124 (integer) 1 => Snowflake\Publications\Domain\Model\Publicationprototypepersistent entity (uid=35323, pid=124) originalId => protected35323 (integer) authors => protected'Oliveira, J.; Pittino, F.; Scheidegger, C.; Fink, S.; Ja
nssen, E. M. -L.' (107 chars) title => protected'Genetic and metabolic diversity of cyanobacteria on the rock–water interfa
ce in mountainous ecosystems' (104 chars) journal => protected'Environmental Science and Technology' (36 chars) year => protected2025 (integer) volume => protected59 (integer) issue => protected'38' (2 chars) startpage => protected'20595' (5 chars) otherpage => protected'20607' (5 chars) categories => protected'metabarcoding; toxin; anabaenopeptin; microcystin; cyanopeptolin; Tintenstri
ch; lichen' (86 chars) description => protected'<em>Tintenstrich</em> communities are specialized lithic biofilms dominated
by free-living cyanobacteria, also occurring in lichen associations, forming
a unique ecological interface between rock environments and aquatic habitat
s in mountainous areas. To better understand their composition and genetic a
nd metabolic potential, we analyzed 207 samples from the Swiss Alps and Jura
Mountains. We determined how key environmental factors shaped cyanobacteria
l abundance, assessed whether these communities harbor genes for toxin biosy
nthesis, characterized their taxonomic composition at the family and genus l
evel, and evaluated the actual occurrence of cyanotoxins and other bioactive
metabolites. Cyanobacterial abundance proved to be influenced by factors su
ch as elevation, exposure, and their interaction with siliceous rock substra
ta. Targeted PCR and Sanger sequencing revealed the presence of toxin-encodi
ng genes, particularly for <em>ndaF</em>/<em>mcyE</em> fragments, which may
encode microcystin and/or nodularin biosynthesis, while specific genes for m
icrocystins, anatoxins, and cylindrospermopsins were rather rare. Metabarcod
ing analysis identified 11 cyanobacterial families, with Chroococcaceae, Nos
tocaceae, and Leptolyngbyaceae being the most abundant. Complementary high-r
esolution mass spectrometry confirmed the occasional presence of nodularins
and microcystins, alongside more frequent detection of other bioactive pepti
des such as anabaenopeptins and cyanopeptolins. Overall, these findings prov
ide the most comprehensive insight to date into <em>Tintenstrich</em>-associ
ated cyanobacteria, underscoring their environmental significance given thei
r genetic and metabolic potential.' (1706 chars) serialnumber => protected'0013-936X' (9 chars) doi => protected'10.1021/acs.est.5c05763' (23 chars) uid => protected35323 (integer) _localizedUid => protected35323 (integer)modified _languageUid => protectedNULL _versionedUid => protected35323 (integer)modified pid => protected124 (integer) 2 => Snowflake\Publications\Domain\Model\Publicationprototypepersistent entity (uid=33271, pid=124) originalId => protected33271 (integer) authors => protected'Wang, X.; Wullschleger, S.; Jones, M.; Reyes, M.; Bossar
t, R.; Pomati, F.; Janssen, E. M. -L.' (138 chars) title => protected'Tracking extensive portfolio of cyanotoxins in five-year lake survey and ide
ntifying indicator metabolites of cyanobacterial taxa' (129 chars) journal => protected'Environmental Science and Technology' (36 chars) year => protected2024 (integer) volume => protected58 (integer) issue => protected'37' (2 chars) startpage => protected'16560' (5 chars) otherpage => protected'16569' (5 chars) categories => protected'microcystin; suspect screening; monitoring; cyanopepetides; harmful algal bl
oom' (79 chars) description => protected'Cyanobacterial blooms require monitoring, as they pose a threat to ecosystem
s and human health, especially by the release of toxins. Along with widely r
eported microcystins, cyanobacteria coproduce other bioactive metabolites; h
owever, information about their dynamics in surface waters is sparse. We inv
estigated dynamics across full bloom successions throughout a five-year lake
monitoring campaign (Greifensee, Switzerland) spanning 150 sampling dates.
We conducted extensive suspect screening of cyanobacterial metabolites using
the database CyanoMetDB. Across all 850 samples, 35 metabolites regularly c
o-occurred. Microcystins were present in 70% of samples, with [d-Asp<sup>3</
sup>,(<em>E</em>)-Dhb<sup>7</sup>]MC-RR reaching concentrations of 70 ng/L.
Anabaenopeptins, meanwhile, were detected in 95% of all samples with concent
rations of Oscillamide Y up to 100-fold higher than microcystins. Based on L
C-MS response and frequency, we identified indicator metabolites exclusively
produced by one of three cyanobacteria isolated from the lake, these being
[d-Asp<sup>3</sup>,(<em>E</em>)-Dhb<sup>7</sup>]MC-RR from <em>Planktothrix<
/em> sp. G2020, Microginin 761B from <em>Microcystis</em> sp. G2011, and Fer
intoic acid B from <em>Microcystis</em> sp. G2020. These indicators showed d
istinct temporal trends and peaking seasons that reflect the variance in eit
her the abundance of the producing cyanobacteria or their toxin production d
ynamics. Our approach demonstrates that selecting high LC-MS response and fr
equent and species-specific indicator metabolites can be advantageous for cy
anobacterial monitoring.' (1620 chars) serialnumber => protected'0013-936X' (9 chars) doi => protected'10.1021/acs.est.4c04813' (23 chars) uid => protected33271 (integer) _localizedUid => protected33271 (integer)modified _languageUid => protectedNULL _versionedUid => protected33271 (integer)modified pid => protected124 (integer) 3 => Snowflake\Publications\Domain\Model\Publicationprototypepersistent entity (uid=33380, pid=124) originalId => protected33380 (integer) authors => protected'Steiner, T.; Schanbacher, F.; Lorenzen, W.; Enke, H.; Ja
nssen, E. M. -L.; Niedermeyer, T. H. J.; Gadem
ann, K.' (164 chars) title => protected'UV–vis absorbance spectra, molar extinction coefficients and circular dich
roism spectra for the two cyanobacterial metabolites anabaenopeptin A and an
abaenopeptin B' (166 chars) journal => protected'Data in Brief' (13 chars) year => protected2024 (integer) volume => protected57 (integer) issue => protected'' (0 chars) startpage => protected'110914 (13 pp.)' (15 chars) otherpage => protected'' (0 chars) categories => protected'cyanobacteria; metabolites; quantitative NMR; UV–vis spectroscopy' (67 chars) description => protected'The UV–vis absorbance spectra, molar extinction coefficients and circular
dichroism spectra, as well as NMR and high resolution tandem mass spectromet
ry spectra were determined for two prominent secondary metabolites from cyan
obacteria, namely anabaenopeptin A and anabaenopeptin B. The compounds were
extracted from the cyanobacterium <em>Planktothrix rubescens</em> CBT929 and
purified by flash chromatography and HPLC. Exact amounts of isolated compou
nds were assessed by quantitative <sup>1</sup>H-NMR with internal calibrant
ethyl 4-(dimethylamino)benzoate in DMSO‑<em>d<sub>6</sub></em> at 298 K wi
th a recycle delay (d1) of 120 s. UV–vis absorbance spectra were recorded
in methanol at room temperature. Molar extinction coefficients were determin
ed at 278 nm as 4190 M<sup>−1</sup> cm<sup>−1</sup> and 2300 M<sup>−1<
/sup> cm<sup>−1</sup> in methanol for anabaenopeptin A and anabaenopeptin
B, respectively. Circular dichroism spectra and secondary fragmentation mass
spectra are also reported.' (1015 chars) serialnumber => protected'2352-3409' (9 chars) doi => protected'10.1016/j.dib.2024.110914' (25 chars) uid => protected33380 (integer) _localizedUid => protected33380 (integer)modified _languageUid => protectedNULL _versionedUid => protected33380 (integer)modified pid => protected124 (integer) 4 => Snowflake\Publications\Domain\Model\Publicationprototypepersistent entity (uid=33003, pid=124) originalId => protected33003 (integer) authors => protected'Torres, M. de A.; Dax, A.; Grand, I.; vom Berg,&nbs
p;C.; Pinto, E.; Janssen, E. M..L.' (127 chars) title => protected'Lethal and behavioral effects of semi-purified microcystins, Micropeptin and
apolar compounds from cyanobacteria on freshwater microcrustacean Thamnocep
halus platyurus' (167 chars) journal => protected'Aquatic Toxicology' (18 chars) year => protected2024 (integer) volume => protected273 (integer) issue => protected'' (0 chars) startpage => protected'106983 (9 pp.)' (14 chars) otherpage => protected'' (0 chars) categories => protected'cyanobacterial metabolites; grazer toxicity; malformation; locomotor behavio
ur; cyanopeptolin; microginin' (105 chars) description => protected'The mass proliferation of cyanobacteria, episodes known as blooms, is a conc
ern worldwide. One of the most critical aspects during these blooms is the p
roduction of toxic secondary metabolites that are not limited to the four cy
anotoxins recognized by the World Health Organization. These metabolites com
prise a wide range of structurally diverse compounds that possess bioactive
functions. Potential human and ecosystem health risks posed by these metabol
ites and co-produced mixtures remain largely unknown. We studied acute letha
l and sublethal effects measured as impaired mobility on the freshwater micr
ocrustaceans Thamnocephalus platyurus for metabolite mixtures from two cyano
bacterial strains, a microcystin (MC) producer and a non-MC producer. Both c
yanobacterial extracts, from the MC-producer and non-MC-producer, caused acu
te toxicity with LC<sub>50</sub> (24 h) values of 0.50 and 2.55 mg<sub>dw_bi
omass</sub>/mL, respectively, and decreased locomotor activity. Evaluating t
he contribution of different cyanopeptides revealed that the Micropeptin-K13
9-dominated fraction from the MC-producer extract contributed significantly
to mortality and locomotor impairment of the microcrustaceans, with potentia
l mixture effect with other cyanopeptolins present in this fraction. In the
non-MC-producer extract, compounds present in the apolar fraction contribute
d mainly to mortality, locomotor impairment, and morphological changes in th
e antennae of the microcrustacean. No lethal or sublethal effects were obser
ved in the fractions dominated by other cyanopetides (Cyanopeptolin 959, Nos
toginin BN741). Our findings contribute to the growing body of research indi
cating that cyanobacterial metabolites beyond traditional cyanotoxins cause
detrimental effects. This underscores the importance of toxicological assess
ments of such compounds, also at sublethal levels.' (1874 chars) serialnumber => protected'0166-445X' (9 chars) doi => protected'10.1016/j.aquatox.2024.106983' (29 chars) uid => protected33003 (integer) _localizedUid => protected33003 (integer)modified _languageUid => protectedNULL _versionedUid => protected33003 (integer)modified pid => protected124 (integer) 5 => Snowflake\Publications\Domain\Model\Publicationprototypepersistent entity (uid=33024, pid=124) originalId => protected33024 (integer) authors => protected'Rougé, V.; von Gunten, U.; Janssen, E. M. L.' (70 chars) title => protected'Reactivity of cyanobacteria metabolites with ozone: multicompound competitio
n kinetics' (86 chars) journal => protected'Environmental Science and Technology' (36 chars) year => protected2024 (integer) volume => protected58 (integer) issue => protected'26' (2 chars) startpage => protected'11802' (5 chars) otherpage => protected'11811' (5 chars) categories => protected'cyanopeptides; planktothrix; microcystis; micropollutant; ozonation; toxins;
microcystin' (88 chars) description => protected'Cyanobacterial blooms occur at increasing frequency and intensity, notably i
n freshwater. This leads to the introduction of complex mixtures of their pr
oducts, i.e., cyano-metabolites, to drinking water treatment plants. To asse
ss the fate of cyano-metabolite mixtures during ozonation, a novel multicomp
ound ozone (O<sub>3</sub>) competition kinetics method was developed. Sixtee
n competitors with known second-order rate constants for their reaction with
. The apparent second-order rate constants (<em>k</em><sub>app,O3</sub>) at
pH 7 were simultaneously determined for 31 cyano-metabolites. <em>k</em><sub
>app,O3</sub> for olefin- and phenol-containing cyano-metabolites were consi
stent with their expected reactivity (0.4–1.7 × 10<sup>6</sup> M<sup>–1
</sup> s<sup>–1</sup>) while <em>k</em><sub>app,O3</sub> for tryptophan- a
nd thioether-containing cyano-metabolites were significantly higher than exp
ected (3.4–7.3 × 10<sup>7</sup> M<sup>–1</sup> s<sup>–1</sup>). Cyano
-metabolites containing these moieties are predicted to be well abated durin
g ozonation. For cyano-metabolites containing heterocycles, <em>k</em><sub>a
tivity of this class of compounds. Due to lower O<sub>3</sub> reactivities,
heterocycle- and aliphatic amine-containing cyano-metabolites may be only pa
rtially degraded by a direct O<sub>3</sub> reaction near circumneutral pH. H
ydroxyl radicals, which are formed during ozonation, may be more important f
or their abatement. This novel multicompound kinetic method allows a high-th
roughput screening of ozonation kinetics.' (1865 chars) serialnumber => protected'0013-936X' (9 chars) doi => protected'10.1021/acs.est.4c02242' (23 chars) uid => protected33024 (integer) _localizedUid => protected33024 (integer)modified _languageUid => protectedNULL _versionedUid => protected33024 (integer)modified pid => protected124 (integer) 6 => Snowflake\Publications\Domain\Model\Publicationprototypepersistent entity (uid=31895, pid=124) originalId => protected31895 (integer) authors => protected'de Almeida Torres, M.; Jones, M. R.; vom Berg, C.; Pinto
, E.; Janssen, E. M. -L.' (120 chars) title => protected'Lethal and sublethal effects towards zebrafish larvae of microcystins and ot
her cyanopeptides produced by cyanobacteria' (119 chars) journal => protected'Aquatic Toxicology' (18 chars) year => protected2023 (integer) volume => protected263 (integer) issue => protected'' (0 chars) startpage => protected'106689 (11 pp.)' (15 chars) otherpage => protected'' (0 chars) categories => protected'cyanobacterial metabolites; fish toxicity; cardiotoxicity; locomotor behavio
ur; cyanopeptolin; microginin' (105 chars) description => protected'Cyanobacterial blooms affect aquatic ecosystems across the globe and one maj
or concern relates to their toxins such as microcystins (MC). Yet, the ecoto
xicological risks, particularly non-lethal effects, associated with other co
-produced secondary metabolites remain mostly unknown. Here, we assessed sur
vival, morphological alterations, swimming behaviour and cardiovascular func
tions of zebrafish (<em>Danio rerio</em>) upon exposure to cyanobacterial ex
tracts of two Brazilian <em>Microcystis</em> strains. We verified that only
MIRS-04 produced MCs and identified other co-produced cyanopeptides also for
the MC non-producer NPCD-01 by LC-HRMS/MS analysis. Both cyanobacterial ext
racts, from the MC-producer and non-producer, caused acute toxicity in zebra
fish with LC<sub>50</sub> values of 0.49 and 0.98 mg<sub>dw_biomass</sub>/mL
, respectively. After exposure to MC-producer extract, additional decreased
locomotor activity was observed. The cyanopeptolin (micropeptin K139) contri
buted 52% of the overall mortality and caused oedemas of the pericardial reg
ion. Oedemas of the pericardial area and prevented hatching were also observ
ed upon exposure to the fraction with high abundance of a microginin (Nostog
inin BN741) in the extract of the MC non-producer. Our results further add t
o the yet sparse understanding of lethal and sublethal effects caused by cya
nobacterial metabolites other than MCs and the need to better understand the
underlying mechanisms of the toxicity. We emphasize the importance of consi
dering mixture toxicity of co-produced metabolites in the ecotoxicological r
isk assessment of cyanobacterial bloom events, given the importance for pred
icting adverse outcomes in fish and other organisms.' (1724 chars) serialnumber => protected'0166-445X' (9 chars) doi => protected'10.1016/j.aquatox.2023.106689' (29 chars) uid => protected31895 (integer) _localizedUid => protected31895 (integer)modified _languageUid => protectedNULL _versionedUid => protected31895 (integer)modified pid => protected124 (integer) 7 => Snowflake\Publications\Domain\Model\Publicationprototypepersistent entity (uid=24660, pid=124) originalId => protected24660 (integer) authors => protected'Jones, M. R.; Janssen, E. M. L.' (56 chars) title => protected'Quantification of multi-class cyanopeptides in Swiss Lakes with automated ex
traction, enrichment and analysis by online-SPE HPLC-HRMS/MS' (136 chars) journal => protected'Chimia' (6 chars) year => protected2022 (integer) volume => protected76 (integer) issue => protected'1-2' (3 chars) startpage => protected'133' (3 chars) otherpage => protected'144' (3 chars) categories => protected'analytical method; cyanobacteria; cyanotoxins; mass spectrometry; natural to
xins' (80 chars) description => protected'The frequency and intensity of cyanobacterial blooms continue to increase in
freshwater systems across the globe. Cyanobacteria can release toxins and s
everal bioactive secondary metabolites and analytical methods are needed to
effectively assess their concentrations in surface waters. Since booms can e
volve rapidly in parts of a lake, high resolution of spatial and temporal sa
mpling increases the complexity of monitoring efforts. Here, we present the
validation of an automated, online-solid phase extraction (SPE) high perform
ance liquid chromatography (HPLC)-high resolution tandem mass spectrometry (
HRMS/MS) method. This online-SPE HPLC-HRMS/MS methods enables quantitative m
onitoring of surface waters for 17 cyanobacterial peptides (cyanopeptides),
spanning 5 distinct cyanopeptide classes, including: microcystins, anabaenop
eptins, nodularins, cyclamides and cyanopeptolins. The method can quantify t
hese cyanopeptides in the low ng/L-range with high accuracy (85-116%) and lo
w relative matrix effects (<25%). We demonstrated its application to Swis
s lake waters (Zürichsee, Hallwilersee, Greifensee), which also highlighted
the value of adding cyanopeptides beyond common microcystins when monitorin
g surface waters for cyanobacteria.' (1251 chars) serialnumber => protected'0009-4293' (9 chars) doi => protected'10.2533/chimia.2022.133' (23 chars) uid => protected24660 (integer) _localizedUid => protected24660 (integer)modified _languageUid => protectedNULL _versionedUid => protected24660 (integer)modified pid => protected124 (integer) 8 => Snowflake\Publications\Domain\Model\Publicationprototypepersistent entity (uid=26097, pid=124) originalId => protected26097 (integer) authors => protected'Pittino, F.; Oliveira, J.; De Almeida Torres, M.; Fink,
S.; Janssen, E. M. L.; Scheidegger, C.' (134 chars) title => protected'Cyanobacteria: extreme environments and toxic metabolites' (57 chars) journal => protected'Chimia' (6 chars) year => protected2022 (integer) volume => protected76 (integer) issue => protected'11' (2 chars) startpage => protected'967' (3 chars) otherpage => protected'969' (3 chars) categories => protected'bioaccumulation; cyanobacteria; cyanotoxins; lichen; water quality' (66 chars) description => protected'Cyanobacteria, also known as blue-green algae, are photosynthetic bacteria t
hat can colonize different habitats, including extreme ones. They are of gre
at interest to the scientific community, especially because of their ability
to produce cyanotoxins: toxic secondary metabolites potentially harmful to
organisms especially when released to surface waters.' (357 chars) serialnumber => protected'0009-4293' (9 chars) doi => protected'10.2533/chimia.2022.967' (23 chars) uid => protected26097 (integer) _localizedUid => protected26097 (integer)modified _languageUid => protectedNULL _versionedUid => protected26097 (integer)modified pid => protected124 (integer) 9 => Snowflake\Publications\Domain\Model\Publicationprototypepersistent entity (uid=23735, pid=124) originalId => protected23735 (integer) authors => protected'van Santen, J. A.; Poynton, E. F.; Iskakova, D.; M
cMann, E.; Alsup, T. A.; Clark, T. N.; Fergusson,
C. H.; Fewer, D. P.; Hughes, A. H.; McCadden,
C. A.; Parra, J.; Soldatou, S.; Rudolf, J. D.; Jans
sen, E. M. -L.; Duncan, K. R.; Linington, R.&n
bsp;G.' (386 chars) title => protected'The natural products atlas 2.0: a database of microbially-derived natural pr
oducts' (82 chars) journal => protected'Nucleic Acids Research' (22 chars) year => protected2022 (integer) volume => protected50 (integer) issue => protected'D1' (2 chars) startpage => protected'D1317' (5 chars) otherpage => protected'D1323' (5 chars) categories => protected'' (0 chars) description => protected'Within the natural products field there is an increasing emphasis on the stu
dy of compounds from microbial sources. This has been fuelled by interest in
the central role that microorganisms play in mediating both interspecies in
teractions and host-microbe relationships. To support the study of natural p
roducts chemistry produced by microorganisms we released the Natural Product
s Atlas, a database of known microbial natural products structures, in 2019.
This paper reports the release of a new version of the database which inclu
des a full RESTful application programming interface (API), a new website fr
amework, and an expanded database that includes 8128 new compounds, bringing
the total to 32 552. In addition to these structural and content changes we
have added full taxonomic descriptions for all microbial taxa and have adde
d chemical ontology terms from both NP Classifier and ClassyFire. We have al
so performed manual curation to review all entries with incomplete configura
tional assignments and have integrated data from external resources, includi
ng CyanoMetDB. Finally, we have improved the user experience by updating the
Overview dashboard and creating a dashboard for taxonomic origin. The datab
ase can be accessed via the new interactive website at https://www.npatlas.o
rg.' (1295 chars) serialnumber => protected'0305-1048' (9 chars) doi => protected'10.1093/nar/gkab941' (19 chars) uid => protected23735 (integer) _localizedUid => protected23735 (integer)modified _languageUid => protectedNULL _versionedUid => protected23735 (integer)modified pid => protected124 (integer) 10 => Snowflake\Publications\Domain\Model\Publicationprototypepersistent entity (uid=23739, pid=124) originalId => protected23739 (integer) authors => protected'Natumi, R.; Dieziger, C.; Janssen, E. M. -L.' (69 chars) title => protected'Cyanobacterial toxins and cyanopeptide transformation kinetics by singlet ox
ygen and pH-dependence in sunlit surface waters' (123 chars) journal => protected'Environmental Science and Technology' (36 chars) year => protected2021 (integer) volume => protected55 (integer) issue => protected'22' (2 chars) startpage => protected'15196' (5 chars) otherpage => protected'15205' (5 chars) categories => protected'cyanopeptide; cyanobacteria; microcystin; phototransformation; anabaenopepti
n; natural toxins; tyrosine; singlet oxygen' (119 chars) description => protected'To assess the risks associated with cyanobacterial blooms, the persistence a
nd fate processes of cyanotoxins and other bioactive cyanobacterial metaboli
tes need to be evaluated. Here, we investigated the reaction with photochemi
cally produced singlet oxygen (<sup>1</sup>O<sub>2</sub>) for 30 cyanopeptid
es synthesized by <em>Dolichospermum flos aquae</em>, including 9 anabaenope
ptins, 18 microcystins, 2 cyanopeptolins, and 1 cyclamide. All compounds wer
e stable in UVA light alone but in the presence of a photosensitizer we obse
rved compound-specific degradation. A strong pH effect on the decay was obse
rved for 18 cyanopeptides that all contained tyrosine or structurally relate
d moieties. We can attribute this effect to the reaction with <sup>1</sup>O<
sub>2</sub> and triplet sensitizer that preferentially react with the deprot
onated form of tyrosine moieties. The contribution of <sup>1</sup>O<sub>2</s
ub> to indirect phototransformation ranged from 12 to 39% and second-order r
ate constants for 9 tyrosine-containing cyanopeptides were assessed. Includi
ng the pH dependence of the reaction and system-independent second-order rat
e constants with <sup>1</sup>O<sub>2</sub> will improve the estimation of ha
lf-lives for multiclass cyanopeptide in surface waters. Our data further ind
icates that naturally occurring triplet sensitizers are likely to oxidize de
protonated tyrosine moieties of cyanopeptides and the specific reactivity an
d its pH dependence needs to be investigated in future studies.' (1507 chars) serialnumber => protected'0013-936X' (9 chars) doi => protected'10.1021/acs.est.1c04194' (23 chars) uid => protected23739 (integer) _localizedUid => protected23739 (integer)modified _languageUid => protectedNULL _versionedUid => protected23739 (integer)modified pid => protected124 (integer) 11 => Snowflake\Publications\Domain\Model\Publicationprototypepersistent entity (uid=22156, pid=124) originalId => protected22156 (integer) authors => protected'Filatova, D.; Jones, M. R.; Haley, J. A.; Núñez,&
nbsp;O.; Farré, M.; Janssen, E. M. -L.' (135 chars) title => protected'Cyanobacteria and their secondary metabolites in three freshwater reservoirs
in the United Kingdom' (98 chars) journal => protected'Environmental Sciences Europe' (29 chars) year => protected2021 (integer) volume => protected33 (integer) issue => protected'' (0 chars) startpage => protected'29 (11 pp.)' (11 chars) otherpage => protected'' (0 chars) categories => protected'cyanotoxin; cyanopeptide; water quality; mass spectrometry; suspect-screenin
g; surface water; natural toxin; emerging contaminant' (129 chars) description => protected'<em>Background:</em> Bloom-forming cyanobacteria occur globally in aquatic e
nvironments. They produce diverse bioactive metabolites, some of which are k
nown to be toxic. The most studied cyanobacterial toxins are microcystins, a
natoxin, and cylindrospermopsin, yet more than 2000 bioactive metabolites ha
ve been identified to date. Data on the occurrence of cyanopeptides other th
an microcystins in surface waters are sparse.<br /><em>Results:</em> We used
a high-performance liquid chromatography–high-resolution tandem mass spe
ctrometry/tandem mass spectrometry (HPLC–HRMS/MS) method to analyse cyanot
oxin and cyanopeptide profiles in raw drinking water collected from three fr
eshwater reservoirs in the United Kingdom. A total of 8 cyanopeptides were i
dentified and quantified using reference standards. A further 20 cyanopeptid
es were identified based on a suspect-screening procedure, with class-equiva
lent quantification. Samples from Ingbirchworth reservoir showed the highest
total cyanopeptide concentrations, reaching 5.8, 61, and 0.8 µg/L in Augu
st, September, and October, respectively. Several classes of cyanopeptides w
ere identified with anabaenopeptins, cyanopeptolins, and microcystins domina
ting in September with 37%, 36%, and 26%, respectively. Samples from Tophill
Low reservoir reached 2.4 µg/L in September, but remained below 0.2 µg/
L in other months. Samples from Embsay reservoir did not exceed 0.1 µg/L.
At Ingbirchworth and Tophill Low, the maximum chlorophyll-a concentrations o
eptide concentrations. These values exceed the World Health Organization’s
guideline levels for relatively low probability of adverse health effects,
which are defined as 10 µg/L chlorophyll-a and 2 × 10<sup>4</sup> cel
ls/mL.<br /><em>Conclusions:</em> This data is the first to present concentr
ations of anabaenopeptin...' (2278 chars) serialnumber => protected'2190-4707' (9 chars) doi => protected'10.1186/s12302-021-00472-4' (26 chars) uid => protected22156 (integer) _localizedUid => protected22156 (integer)modified _languageUid => protectedNULL _versionedUid => protected22156 (integer)modified pid => protected124 (integer) 12 => Snowflake\Publications\Domain\Model\Publicationprototypepersistent entity (uid=22310, pid=124) originalId => protected22310 (integer) authors => protected'Jones, M. R.; Pinto, E.; Torres, M. A.; Dörr, 
;F.; Mazur-Marzec, H.; Szubert, K.; Tartaglione, L.; Dell'Ave
rsano, C.; Miles, C. O.; Beach, D. G.; McCarron,&nb
sp;P.; Sivonen, K.; Fewer, D. P.; Jokela, J.; Janssen,&n
bsp;E. M. -L.' (327 chars) title => protected'CyanoMetDB, a comprehensive public database of secondary metabolites from cy
anobacteria' (87 chars) journal => protected'Water Research' (14 chars) year => protected2021 (integer) volume => protected196 (integer) issue => protected'' (0 chars) startpage => protected'117017 (12 pp.)' (15 chars) otherpage => protected'' (0 chars) categories => protected'cyanobacteria; secondary metabolite; database; toxin; cyanopeptide; CyanoMet
DB' (78 chars) description => protected'Harmful cyanobacterial blooms, which frequently contain toxic secondary meta
bolites, are reported in aquatic environments around the world. More than tw
o thousand cyanobacterial secondary metabolites have been reported from dive
rse sources over the past fifty years. A comprehensive, publically-accessibl
e database detailing these secondary metabolites would facilitate research i
nto their occurrence, functions and toxicological risks. To address this nee
d we created CyanoMetDB, a highly curated, flat-file, openly-accessible data
base of cyanobacterial secondary metabolites collated from 850 peer-reviewed
articles published between 1967 and 2020. CyanoMetDB contains 2010 cyanobac
terial metabolites and 99 structurally related compounds. This has nearly do
ubled the number of entries with complete literature metadata and structural
composition information compared to previously available open access databa
ses. The dataset includes microcytsins, cyanopeptolins, other depsipeptides,
anabaenopeptins, microginins, aeruginosins, cyclamides, cryptophycins, saxi
toxins, spumigins, microviridins, and anatoxins among other metabolite class
es. A comprehensive database dedicated to cyanobacterial secondary metabolit
es facilitates: (1) the detection and dereplication of known cyanobacterial
toxins and secondary metabolites; (2) the identification of novel natural pr
oducts from cyanobacteria; (3) research on biosynthesis of cyanobacterial se
condary metabolites, including substructure searches; and (4) the investigat
ion of their abundance, persistence, and toxicity in natural environments.' (1594 chars) serialnumber => protected'0043-1354' (9 chars) doi => protected'10.1016/j.watres.2021.117017' (28 chars) uid => protected22310 (integer) _localizedUid => protected22310 (integer)modified _languageUid => protectedNULL _versionedUid => protected22310 (integer)modified pid => protected124 (integer) 13 => Snowflake\Publications\Domain\Model\Publicationprototypepersistent entity (uid=22112, pid=124) originalId => protected22112 (integer) authors => protected'Natumi, R.; Marcotullio, S.; Janssen, E. M. -L.' (72 chars) title => protected'Phototransformation kinetics of cyanobacterial toxins and secondary metaboli
tes in surface waters' (97 chars) journal => protected'Environmental Sciences Europe' (29 chars) year => protected2021 (integer) volume => protected33 (integer) issue => protected'' (0 chars) startpage => protected'26 (13 pp.)' (11 chars) otherpage => protected'' (0 chars) categories => protected'cyanopeptide; cyanobacteria; microcystin; phototransformation; anabaenopepti
n; natural toxins; surface water; water quality; degradation' (136 chars) description => protected'<em>Background:</em> Cyanobacteria and their toxins occur in high concentrat
ions during the so-called bloom events in surface waters. To be able to asse
ss the risks associated with cyanobacterial blooms, we need to understand th
e persistence and fate processes of these toxins and other bioactive metabol
ites. In this study, we investigated the photochemical fate of 54 cyanopepti
des extracted from two strains of <em>Microcystis aeruginosa</em> (PCC7806 a
nd UV006), <em>Planktothrix rubescens,</em> and <em>Dolichospermum flos aqua
e</em>. We determined half-lives during sunlight exposure in lake water and
inspected the effect of pH on transformation kinetics for 27 microcystins, 8
anabaenopeptins, 14 cyanopeptolins, 2 cyclamides, and 3 aeruginosins.<br />
<em>Results:</em> For cyanopeptides from <em>D. flos aquae</em> and <em>P. r
ubescens,</em> we observed the highest removal of 28 and 26%, respectively,
after 3-h sunlight exposure. Most cyanopeptides produced by the two <em>M. a
eruginosa</em> strains were rather persistent with only up to 3% removal. Th
e more reactive cyanopeptides contained amino acids known to undergo phototr
ansformation, including methionine and tyrosine moieties or their derivative
s. Photochemical half-lives of 14 tyrosine-containing cyanopeptides decrease
d by one order of magnitude from nearly persistent conditions at pH 7 (half-
life > 70 h) to shorter half-lives at pH 10 (< 10 h).<br /><em
>Conclusions:</em> More work is needed to distinguish the contribution of di
fferent photochemical reaction pathways including the contributions to the p
H effect. To the best of our knowledge, this is the first assessment of tran
sformation kinetics of such a wide range of cyanopeptides. The abundant and
persistent cyanopeptides that have not been studied in detail yet should be
prioritized for the evaluation of their ecosystem and human health risks and
for their abatement during drinking water treatment.' (1953 chars) serialnumber => protected'2190-4707' (9 chars) doi => protected'10.1186/s12302-021-00465-3' (26 chars) uid => protected22112 (integer) _localizedUid => protected22112 (integer)modified _languageUid => protectedNULL _versionedUid => protected22112 (integer)modified pid => protected124 (integer) 14 => Snowflake\Publications\Domain\Model\Publicationprototypepersistent entity (uid=20660, pid=124) originalId => protected20660 (integer) authors => protected'Natumi, R.; Janssen, E. M. -L.' (50 chars) title => protected'Cyanopeptide co-production dynamics beyond mirocystins and effects of growth
stages and nutrient availability' (109 chars) journal => protected'Environmental Science and Technology' (36 chars) year => protected2020 (integer) volume => protected54 (integer) issue => protected'10' (2 chars) startpage => protected'6063' (4 chars) otherpage => protected'6072' (4 chars) categories => protected'' (0 chars) description => protected'Intensified cyanobacterial bloom events are of increasing global concern bec
ause of adverse effects associated with the release of bioactive compounds,
including toxic cyanopeptides. Cyanobacteria can produce a variety of cyanop
eptides, yet our knowledge about their abundance and co-production remains l
imited. We applied a suspect-screening approach, including 700 structurally
known cyanopeptides, and identified 11 cyanopeptides in <em>Microcystis aeru
ginosa</em> and 17 in <em>Dolichospermum flos-aquae</em>. Total cyanopeptide
concentrations ranged from high nmol to μmol g<sub>dry</sub><sup>-1</sup>
with slightly higher cell quotas in the mid-exponential growth phase. Relati
ve cyanopeptide profiles were unchanged throughout the growth cycle. We demo
nstrate that quantification based on microcystin-LR equivalents can introduc
e an error of up to 6-fold and recommend a class-equivalent approach instead
. In <em>M. aeruginosa</em>, rarely studied cyclamides dominated (>80%) o
ver cyanopeptolins and microcystins. While all nutrient reductions caused le
ss growth, only lowering phosphorous and micronutrients reduced cyanopeptide
production by <em>M. aeruginosa</em>. Similar trends were observed for <em>
D. flos-aquae</em> and only lowering nitrogen decreased cyanopeptide product
ion while the relative abundance of individual cyanopeptides remained stable
. The synchronized production of other cyanopeptides along with microcystins
emphasizes the need to make them available as reference standards to encour
age more studies on their occurrence in blooms, persistence, and potential t
oxicity.' (1604 chars) serialnumber => protected'0013-936X' (9 chars) doi => protected'10.1021/acs.est.9b07334' (23 chars) uid => protected20660 (integer) _localizedUid => protected20660 (integer)modified _languageUid => protectedNULL _versionedUid => protected20660 (integer)modified pid => protected124 (integer) 15 => Snowflake\Publications\Domain\Model\Publicationprototypepersistent entity (uid=20604, pid=124) originalId => protected20604 (integer) authors => protected'Egli, C. M.; Natumi, R. S.; Jones, M. R.; Jans
sen, E. M. -L.' (105 chars) title => protected'Inhibition of extracellular enzymes exposed to cyanopeptides' (60 chars) journal => protected'Chimia' (6 chars) year => protected2020 (integer) volume => protected74 (integer) issue => protected'3' (1 chars) startpage => protected'122' (3 chars) otherpage => protected'128' (3 chars) categories => protected'aquatic enzymes; biogeochemical cycling; cyanobacteria; harmful algae bloom;
microbial loop' (91 chars) description => protected'Harmful cyanobacterial blooms in freshwater ecosystems produce bioactive sec
ondary metabolites including cyanopeptides that pose ecological and human he
alth risks. Only adverse effects of one class of cyanopeptides, microcystins
, have been studied extensively and have consequently been included in water
quality assessments. Inhibition is a commonly observed effect for enzymes e
xposed to cyanopeptides and has mostly been investigated for human biologica
lly relevant model enzymes. Here, we investigated the inhibition of ubiquito
us aquatic enzymes by cyanobacterial metabolites. Hydrolytic enzymes are uti
lized in the metabolism of aquatic organisms and extracellularly by heterotr
ophic bacteria to obtain assimilable substrates. The ubiquitous occurrence o
f hydrolytic enzymes leads to the co-occurrence with cyanopeptides especiall
y during cyanobacterial blooms. Bacterial leucine aminopeptidase and alkalin
e phosphatase were exposed to cyanopeptide extracts of different cyanobacter
ial strains (<em>Microcystis aeruginosa </em> wild type and microcystin-free
mutant,<em> Planktothrix rubescens</em>) and purified cyanopeptides. We obs
erved inhibition of aminopeptidase and phosphatase upon exposure, especially
to the apolar fractions of the cyanobacterial extracts. Exposure to the dom
inant cyanopeptides in these extracts confirmed that purified microcystins,
aerucyclamide A and cyanopeptolin A inhibit the aminopeptidase in the low mg
L<sup>-1</sup> range while the phosphatase was less affected. Inhibition of
aquatic enzymes can reduce the turnover of nutrients and carbon substrates
and may also impair metabolic functions of grazing organisms.' (1657 chars) serialnumber => protected'0009-4293' (9 chars) doi => protected'10.2533/chimia.2020.122' (23 chars) uid => protected20604 (integer) _localizedUid => protected20604 (integer)modified _languageUid => protectedNULL _versionedUid => protected20604 (integer)modified pid => protected124 (integer) 16 => Snowflake\Publications\Domain\Model\Publicationprototypepersistent entity (uid=18207, pid=124) originalId => protected18207 (integer) authors => protected'Janssen, E. M. -L.' (33 chars) title => protected'Cyanobacterial peptides beyond microcystins – a review on co-occurrence, t
oxicity, and challenges for risk assessment' (119 chars) journal => protected'Water Research' (14 chars) year => protected2019 (integer) volume => protected151 (integer) issue => protected'' (0 chars) startpage => protected'488' (3 chars) otherpage => protected'499' (3 chars) categories => protected'harmful algal bloom; cyanobacteria; toxin; risk assessment; ecotoxicology; h
uman health' (87 chars) description => protected'Cyanobacterial bloom events that produce natural toxins occur in freshwaters
across the globe, yet the potential risk of many cyanobacterial metabolites
remains mostly unknown. Only microcystins, one class of cyanopeptides, have
been studied intensively and the wealth of evidence regarding exposure conc
entrations and toxicity led to their inclusion in risk management frameworks
for water quality. However, cyanobacteria produce an incredible diversity o
f hundreds of cyanopeptides beyond the class of microcystins. The question a
rises, whether the other cyanopeptides are in fact of no human and ecologica
l concern or whether these compounds merely received (too) little attention
thus far. Current observations suggest that an assessment of their (eco)toxi
cological risk is indeed relevant: First, other cyanopeptides, including cya
nopeptolins and anabaenopeptins, can occur just as frequently and at similar
nanomolar concentrations as microcystins in surface waters. Second, cyanope
ptolins, anabaenopeptins, aeruginosins and microginins inhibit proteases in
the nanomolar range, in contrast to protein phosphatase inhibition by microc
ystins. Cyanopeptolins, aeruginosins, and aerucyclamide also show toxicity a
gainst grazers in the micromolar range comparable to microcystins. The key c
hallenge for a comprehensive risk assessment of cyanopeptides remains their
large structural diversity, lack of reference standards, and high analytical
requirements for identification and quantification. One way forward would b
e a prevalence study to identify the priority candidates of tentatively abun
dant, persistent, and toxic cyanopeptides to make comprehensive risk assessm
ents more manageable.' (1693 chars) serialnumber => protected'0043-1354' (9 chars) doi => protected'10.1016/j.watres.2018.12.048' (28 chars) uid => protected18207 (integer) _localizedUid => protected18207 (integer)modified _languageUid => protectedNULL _versionedUid => protected18207 (integer)modified pid => protected124 (integer)
Biotransformation dynamics and products of cyanobacterial secondary metabolites in surface waters
Genetic and metabolic diversity of cyanobacteria on the rock–water interface in mountainous ecosystems
Tracking extensive portfolio of cyanotoxins in five-year lake survey and identifying indicator metabolites of cyanobacterial taxa
UV–vis absorbance spectra, molar extinction coefficients and circular dichroism spectra for the two cyanobacterial metabolites anabaenopeptin A and anabaenopeptin B
Lethal and behavioral effects of semi-purified microcystins, Micropeptin and apolar compounds from cyanobacteria on freshwater microcrustacean Thamnocephalus platyurus
Reactivity of cyanobacteria metabolites with ozone: multicompound competition kinetics
Lethal and sublethal effects towards zebrafish larvae of microcystins and other cyanopeptides produced by cyanobacteria
Quantification of multi-class cyanopeptides in Swiss Lakes with automated extraction, enrichment and analysis by online-SPE HPLC-HRMS/MS
Cyanobacteria: extreme environments and toxic metabolites
The natural products atlas 2.0: a database of microbially-derived natural products
Cyanobacterial toxins and cyanopeptide transformation kinetics by singlet oxygen and pH-dependence in sunlit surface waters
Cyanobacteria and their secondary metabolites in three freshwater reservoirs in the United Kingdom
Results: We used a high-performance liquid chromatography–high-resolution tandem mass spectrometry/tandem mass spectrometry (HPLC–HRMS/MS) method to analyse cyanotoxin and cyanopeptide profiles in raw drinking water collected from three freshwater reservoirs in the United Kingdom. A total of 8 cyanopeptides were identified and quantified using reference standards. A further 20 cyanopeptides were identified based on a suspect-screening procedure, with class-equivalent quantification. Samples from Ingbirchworth reservoir showed the highest total cyanopeptide concentrations, reaching 5.8, 61, and 0.8 µg/L in August, September, and October, respectively. Several classes of cyanopeptides were identified with anabaenopeptins, cyanopeptolins, and microcystins dominating in September with 37%, 36%, and 26%, respectively. Samples from Tophill Low reservoir reached 2.4 µg/L in September, but remained below 0.2 µg/L in other months. Samples from Embsay reservoir did not exceed 0.1 µg/L. At Ingbirchworth and Tophill Low, the maximum chlorophyll-a concentrations of 37 µg/L and 22 µg/L, respectively, and cyanobacterial count of 6 × 104 cells/mL were observed at, or a few days after, peak cyanopeptide concentrations. These values exceed the World Health Organization’s guideline levels for relatively low probability of adverse health effects, which are defined as 10 µg/L chlorophyll-a and 2 × 104 cells/mL.
Conclusions: This data is the first to present concentrations of anabaenopeptins, cyanopeptolins, aeruginosins, and microginins, along with microcystins, in U.K. reservoirs. A better understanding of those cyanopeptides that are abundant in drinking water reservoirs can inform future monitoring and studies on abatement efficiency during water treatment.
CyanoMetDB, a comprehensive public database of secondary metabolites from cyanobacteria
Phototransformation kinetics of cyanobacterial toxins and secondary metabolites in surface waters
Results: For cyanopeptides from D. flos aquae and P. rubescens, we observed the highest removal of 28 and 26%, respectively, after 3-h sunlight exposure. Most cyanopeptides produced by the two M. aeruginosa strains were rather persistent with only up to 3% removal. The more reactive cyanopeptides contained amino acids known to undergo phototransformation, including methionine and tyrosine moieties or their derivatives. Photochemical half-lives of 14 tyrosine-containing cyanopeptides decreased by one order of magnitude from nearly persistent conditions at pH 7 (half-life > 70 h) to shorter half-lives at pH 10 (< 10 h).
Conclusions: More work is needed to distinguish the contribution of different photochemical reaction pathways including the contributions to the pH effect. To the best of our knowledge, this is the first assessment of transformation kinetics of such a wide range of cyanopeptides. The abundant and persistent cyanopeptides that have not been studied in detail yet should be prioritized for the evaluation of their ecosystem and human health risks and for their abatement during drinking water treatment.

