Archive detail
Keeping track of fragrances
March 14, 2019 |
Fragrances are added in a wide variety of consumer products – cosmetics, detergents, cleaning agents, and air fresheners. If incompletely eliminated in wastewater treatment plants, they can end up in rivers and lakes. Companies are therefore required to perform an environmental risk assessment before fragrance compounds are used in final products. One important test parameter is the accumulation of substances in fish. However, the properties of fragrances make them difficult to test: some may readily stick to surfaces and they are by design volatile so that we can smell them. “This means,” says biologist Hannah Schug, PhD student in the Environmental Toxicology department led by Kristin Schirmer, “that, during the experiments, fragrances virtually dissipate.”
An intestinal epithelial cell line serving as a barrier
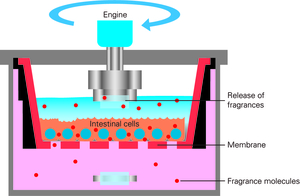
For this reason, Hannah Schug designed a novel test system in collaboration with a leading fragrance company. The device – the size of a small Rubik’s cube – has an inner surface made of polished-mirror stainless steel to reduce losses due to the compounds’ stickiness, and an air-tight lid to minimize evaporation. The system, called TransFEr chamber, consists of an upper and a lower compartment separated by a cellular barrier. This barrier is comprised of fish intestinal epithelial cells representing the intestinal epithelium as a “gatekeeper” in fish. The exposure solution is added to the upper compartment, and measurements are made in the lower compartment to determine the rate of transfer across the intestinal cells. The results indicate to what extent compounds – in this case fragrances – can be absorbed by the intestine to be accumulated or to be transformed in fish.
Potential benefits for pharmaceutical research
As well as being useful for environmental toxicologists, the device developed by Hannah Schug is likely to prove valuable in the future in other fields of research – for example, in human toxicology and pharmacology. Pharmaceutical companies already use human cell lines to determine the intestinal absorption of active medical substances. However, when such substances have challenging properties similar to those of fragrances, they are difficult to test. Schug explains: “With our device, this would now be possible. Whenever it would be demonstrated that a certain substance is unable to cross the human intestinal barrier in the cell-based TransFEr system, one could recommend that animal tests may not even be needed anymore.” Cell-based assays thus have the potential to reduce and perhaps even replace animal experiments in environmental and medical research.
Research efforts to avoid animal experiments
Science and regulation relies on animal experiments to determine the toxicity and accumulation potential of chemical substances. The team led by Kristin Schirmer is working on alternatives to animal testing by developing experimental and theoretical tools designed to reduce or even replace animal experiments for environmental risk assessment in the future.