Archive detail
Dead or alive?
October 30, 2018 |
The composition of human intestinal flora has a significant influence on the immune system and digestion. In addition, changes in intestinal flora – also known as gut microbiota – have been observed to occur in the course of various intestinal diseases.
Researchers are therefore investigating how microorganisms from the human gut could be used for therapeutic purposes. At the ETH Zurich Institute of Food, Nutrition and Health (IFNH), for example, doctoral student Lea Bircher’s recently published dissertation focused on the preservation of gut microbes. If these live microorganisms are to be used in medicine or as dietary supplements, they must not be damaged during storage. Bircher selected six functionally diverse bacterial strains and studied how well they survived storage for three months in a cryopreserved (-80°C) or freeze-dried state, with various protective agents being used to minimize damage.
Knowledge transfer in the area of laboratory methods
Frederik Hammes of Eawag’s Environmental Microbiology department supported this research project with his expertise in flow cytometry – an analytical method that allows microbial cells in drinking water to be quantified more rapidly than is possible with the plate count method or molecular biological techniques.
Clarissa Schwab, who supervised Bircher’s doctoral research at the IFNH, explains: “Flow cytometry wasn’t completely new to us, but as it hadn’t been used in our lab for some years, we were glad to be able to draw on Eawag’s know-how.”
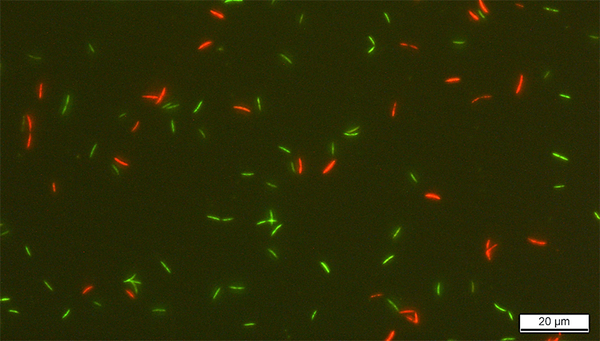
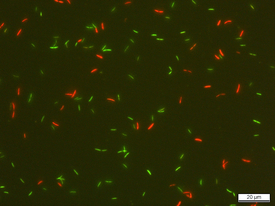
Frederik Hammes has been using flow cytometry in drinking and wastewater analysis for many years and has considerable experience with various methods of assessing the viability of bacteria in water samples. Bircher discussed with Hammes which of these methods would be best suited for assessing the viability of gut bacteria after preservation. They soon decided that the bacteria should be stained with SYBR Green I and propidium iodide (SGPI), which makes it possible to evaluate the integrity of the bacterial membrane: in flow cytometric analyses, intact bacteria appear green, while red fluorescence indicates membrane damage.
Underlying this decision was the fact that Eawag is particularly familiar with the SGPI staining protocol – the method was optimized for flow cytometry by Frederik Hammes’ group and was standardized in 2016. Test runs in the laboratory at Eawag confirmed its suitability for analysing the gut bacteria selected for the project. After learning the procedure at Eawag, Bircher carried out the flow cytometric analyses independently at the ETH labs.
Positive effects of protectants
To assess the survival of the probiotics, Bircher examined cultures of the various strains before and after preservation. The SGPI method yielded valuable additional information on the viability of the samples. The addition of protectants such as sucrose, inulin or glycerol was found to enhance – sometimes markedly – the viability of the bacteria. However, individual protectants are not equally effective with all bacteria and preservation processes.
All the bacteria selected by Bircher are regarded as particularly promising “next-generation” probiotics. However, numerous further tests will be required before these candidates are ready to enter the market.
Where it all began
The collaboration between Clarissa Schwab and Frederik Hammes dates back to an Eawag conference held in 2015. Entitled “How Dead Is Dead?”, this event explored the limits of bacterial life: when is a bacterial cell dead, and how can this be determined? Bacteria can enter a resting state in which their metabolic and physiological activity is so low as to be no longer detectable. Can these cells be “resuscitated”? These questions have not yet been definitively answered – either in medicine or in drinking water microbiology.
Lea Bircher's research was funded by the Swiss National Science Foundation (Sinergia 35150) and ETH Zurich.