Archive detail
Tracking down the smallest particles
June 19, 2018 |
They can make plastics stronger, block UV rays, reduce sweat odour in t-shirts or ensure better water-droplet formation: artificial nanoparticles are used in a wide range of products. They range from one to a hundred nanometres in size – even a hair has a diameter of c. 10,000 nanometres. They are hard to grasp, which is perhaps the reason for their often-dubious reputation among the general population. But, these tiny particles are not just a human invention. Natural nanoparticles are found in volcanic ash or in the smoke from wood fires, and biological and chemical particles, such as fat droplets in milk or small proteins in the blood, are also on a nanoscale.
Transformation in wastewater treatment plants
Artificially produced nanoparticles can enter the environment during the entire life cycle of a product. How the particles then behave or what effects they have are questions which researchers all over the world grapple with. One of these researchers is Ralf Kägi, an expert on nanoparticles at Eawag. In a study published in 2017, for example, he and his team investigated the transformation of copper oxide nanoparticles in wastewater. These are used in conductors and circuits and in wood impregnation. It is assumed that copper oxide is transformed into copper sulphide in wastewater treatment plants and thereby loses most of its toxicity. Within minutes, in fact, copper particles are converted into covellite, a copper sulphide. This mineral is frequently found in nature as a thin film on rocks. As Kägi states: “In this way, the dissolved copper ions are bound together and the particles lose their nano-specific properties”.
Large quantities land in sewage sludge
Other nanoparticles are also quickly transformed in wastewater treatment plants and 95% to 99% end up in sewage sludge. “These are incinerated and deposited, and, therefore, the artificial nanoparticles are no longer in circulation”, says Kägi. He and his team have investigated the risks of nanomaterials for humans and the environment in a number of studies. “We started with silver, which is used mainly in textiles. We have also looked at copper, zinc, titanium and cerium nanoparticles in pilot plants at Eawag, simulating real-world conditions”, he explains. The latter is used in abrasive powders and glass lenses, while titanium is an ingredient in sunscreens and boats are painted with copper. The results are the same: “Wastewater plants transform the reactive nanoparticles relatively quickly into less harmful particles which can then be efficiently removed from wastewater”.
Ecotoxicology investigates biological reactions
Fundamental processes are the focus in the Environmental Toxicology department, under the leadership of Kristin Schirmer. “We are investigating in particular the influence of frequently used metallic nanoparticles on living organisms at different ecological levels of organisation: from algae to biofilms, cell lines, and embryos up to fish”, explains Ecotoxicologist Ahmed Tlili. These investigations take place in the laboratory. Although a range of effects appear with very low concentrations, as a rule the concentrations are higher than those in the environment.
The Ecotoxicologists have investigated, for example, how well the alga Chlamydomonas reinhardtii takes up cerium and cerium oxide nanoparticles. These are used as additives, for instance, in ceramic tooth fillings, in the coatings of self-cleaning ovens and as opacifying agents in special glass, such as windshields. In their study, the researchers compared how the wild type of the alga and a mutant without a cell wall absorb the cerium and cerium dioxide nanoparticles. In the case of cerium dioxide there is no difference, but the dissolved cerium is better absorbed by the mutant. The researchers, thus, assume that cellular cerium adheres strongly to the cell wall of the wild type, and that less is absorbed.
“Algae often cannot take up nanoparticles”, confirms Ahmed Tlili. “But if metallic components are dissolved in water, these can have an effect on algae and bacteria. These are indirectly affected by the nanoparticles”. Biofilms, on the other hand, do react to nanoparticles. “Groups of microbiological organisms show less growth and the respiration rate sinks. This results in less food being available for higher-level organisms”, explains Tlili.
Differentiating between artificial and natural sources
Thanks to comprehensive research in various areas, it is now better known how common nanoparticles land in the environment and where they finally end up. In addition, a large number of analytical methods are available today, which make it possible, for example, to distinguish between artificial and natural nanoparticles. One example is titanium dioxide, that is used in sunscreen, and that is washed off the skin when bathing. Filters in swimming pools remove nearly all the titanium from water. In natural bathing locations, such as lakes and rivers, however, there are no such filters. But, titanium-containing nanoparticles also occur naturally – in bodies of water at a concentration of c. 5,000 particles per millilitre.
In a recently published study, researchers at the University of Vienna, Eawag and the Swedish University of Agricultural Sciences cooperated in an investigation on whether and how much titanium dioxide in sunscreens ends up in lakes and rivers during bathing and is later deposited in sediment. They drew on the fact that natural titanium particles are often found with iron, manganese, lead or aluminium, while in the case of artificial titanium nanoparticles, no such typical ‘background’ is known. “This is useful for differentiating the sources of the particles”, says Kägi. The researchers determined that in the Danube, the titanium oxide concentration increases during the bathing season. The increase resulting from sunscreen, however, was much less than the naturally occurring material. It is also possible to differentiate artificial cerium oxide from the naturally occurring nanomaterial. Kägi explains: “Natural cerium oxide generally occurs with lanthanum, whereas the artificial cerium oxide does not. This fact makes differentiation possible”.
No acute effects in natural conditions
The progress in nano research over the last 10 years and where it should go were the topics of a meeting of international experts, under the leadership of Empa and Eawag, on Monte Verità in March 2017. They noted that the effects of nanoparticles on diverse organisms can be observed after relatively brief periods in laboratory experiments. However, these occur above all at high concentrations, and cannot be observed in the environment. “Such laboratory experiments are important to understand basic processes. But, they are often far removed from reality”, says Kägi. “Under realistic conditions, we see no acute effects on organisms and ecosystems – provided that the nanomaterials do not consist of toxic materials, such as heavy metals”.
Research focus shifts
In the future, Eawag researchers want to concentrate on the many open questions that remain: What chronic effects can artificial nanoparticles cause? How do living communities and ecosystems react when exposed over the long term to a combination of different nanomaterials? What happens to nanomaterials when they are incinerated? “We want to use the great amount of existing data to improve risk modelling”, adds Tlili. “In addition, we want to do less research on original nanomaterials and rather concentrate on particles that have aged in the environment”. This is also true for the research plans of Kägi’s group, which is planning to investigate the transformation of cerium dioxide nanoparticles during the wastewater treatment process, as well as during the treatment and incineration of sludge. Furthermore, the researchers wish to determine if the nanoparticles from the deposited ashes of sludge become active again.
Publications
Nowack, B., 2009. The behavior and effects of nanoparticles in the environment. Environ. Pollut. 157, 1063–1064.
Photos
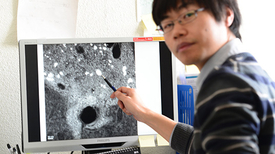
Ecotoxicologist Xiaomei Li points to nanoparticles taken up by cells.
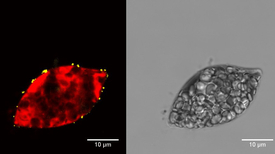
Euglena gracilis, after the alga has been exposed for two hours to fluorescent nanoparticles. Fluorescent particles (left, yellow) are found on the outside of the algae cell, the inside of which fluoresces red (compared to the appearance in transmission microscopy on the right). (Source: Eawag, Xiaomei Li)

In experiments at Eawag, researchers test methods for analysing artificial titanium dioxide nanoparticles from sunscreens in water.

After the bath, researchers take water samples in order to analyse artificial titanium dioxide nanoparticles from sunscreens.

